Go back to the overview of our research and insulin resistance
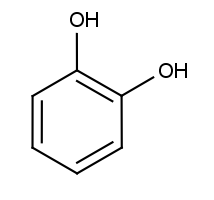
In the interventive arm of our research program, the Laboratory for Evaluation of Pharmacological Agents in Remediation of Disease (LEoPARD) is exploring bioactive agents capable of reversing insulin resistance, which would potentially act as a therapeutic against diseases caused by insulin resistance such as type 2 diabetes. While there are currently antidiabetic drugs in common use, they are not without their issues. The globally-recognized “gold standard” drug, metformin a.k.a. Glucophage, is postulated to lower blood glucose by activating exercise/catabolic signaling pathways in cells — namely the 5′ adenosine monophosphate-activated protein kinase (AMPK) pathway (Hawley et al., 2002). While metformin is widely-lauded as not only an efficacious antidiabetic — but also a “wonderdrug” with astonishing prospective uses ranging to anti-cancer potential — it entails some risks, particularly in our increasingly-aging population. These may include neurodegeneration, muscular atrophy, hypoglycemia, and gastrointestinal distress — as summarized in our 2021 PharmaNutrition review (Knezevic et al., 2021). In said review, we proposed an attractive nutraceutical alternative to metformin: catechol-containing compounds. Catechol — the 1,2-dihydroxybenzene chemical structure seen below — is present as a structural moiety in a variety of natural products associated with potential reversal of insulin resistance or sensitization of the IRSP — such as the catechins (a prominent ingredient in teas; Yan et al., 2012).
In the time since our 2021 PharmaNutrition review was published, we have expanded our characterization of potential bioactive agents to include guaiacol (a methylated form of catechol; seen below), and a variety of nutraceuticals which contain the guaiacol moiety — including the curcuminoids (such as curcumin, the active ingredient in turmeric; Hodaei et al., 2019), and the vanilloid family which contains the capsaicins (the pungent active ingredient in spicy foods; Panchal et al., 2018).
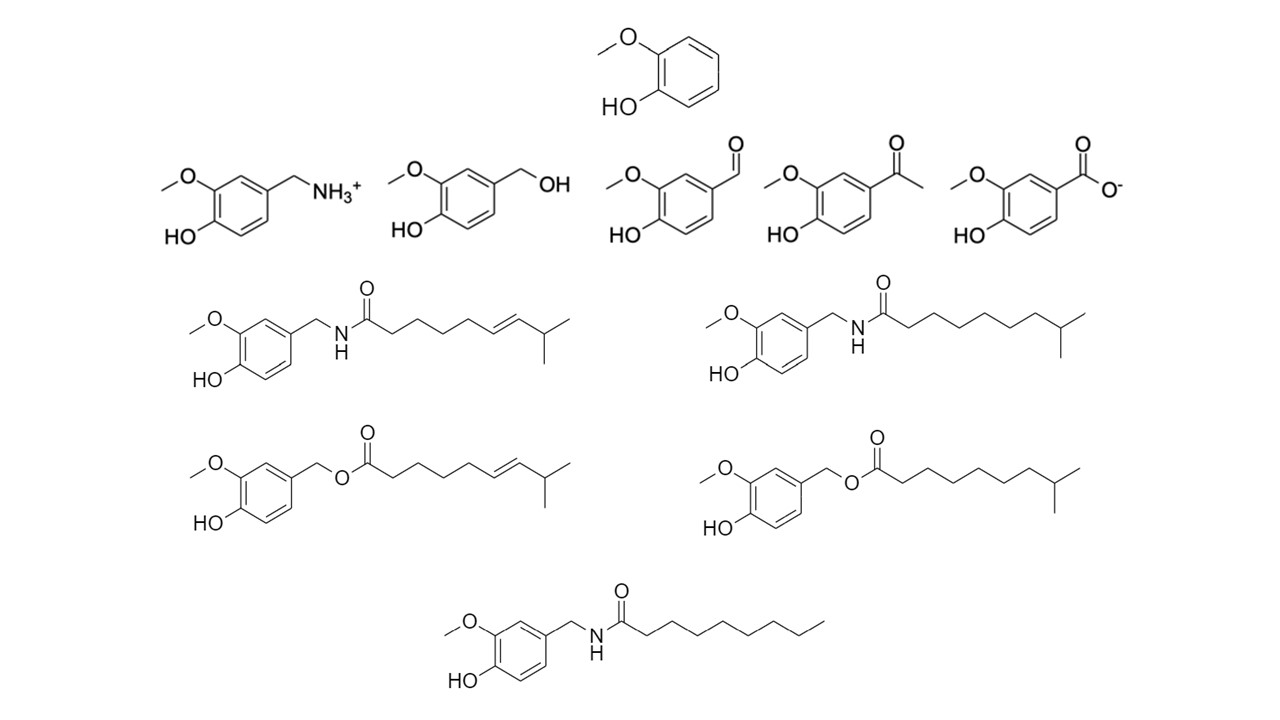
The image above shows depicts guaiacol and the vanilloids under study in our lab. First row: guaiacol; second row (l-r): vanillylamine, vanillyl alcohol, vanillin, acetovanillone, vanillate; third row (l-r): capsaicin, dihydrocapsaicin; fourth row (l-r): capsiate, dihydrocapsiate; fifth row: nonivamide.
Our aims, broadly speaking, are to characterize the molecular mechanisms by which the bioactive compounds listed here — catechol, guaiacol, vanilloids, capsaicins, capsaicinoids, and so forth — might sensitize cellular signal transduction of the insulin receptor signaling pathway, and thereby reverse insulin resistance.
Go back to the overview of our research and insulin resistance

