Overview
The Laboratory for Evaluation of Pharmacological Agents in Remediation of Disease (LEoPARD) is a biochemistry-focused research group focused on the insulin receptor signaling pathway (IRSP), which is arguably the most important — and most complex — signal transduction pathway responsible for regulating the metabolism and physiology of animal cells. The most recognizable molecular pathology arising from IRSP dysfunction is type 2 diabetes; however, insulin resistance (the failure of cells to respond to physiologically normal insulin levels) is also implicated in Alzheimer’s dementia, as well as various cancers (both as a cause and effect), among other diseases. Projects in our lab fall into two “arms” — with elegant overlap between active research projects — with one arm focusing on i) reversing insulin resistance in cells where it has already developed, and the other focusing on ii) early detection of insulin resistance before it develops.
i) The interventive arm — reversal of insulin resistance. We are currently using a molecular pharmacology approach to explore and characterize a family of naturally-occurring chemical compounds known as vanilloids — which are naturally present in various foods associated with ameliorating type 2 diabetes and metabolic syndrome, such as spicy foods, antioxidant roots, and teas.
ii) The preventive arm — early detection of insulin resistance.
What is insulin resistance?
The insulin receptor signaling pathway (IRSP) is arguably one of the most important growth and proliferation signaling pathways responsible for regulating animal cells (Haeusler et al., 2018). It is most recognizable as an insulin-responsive pathway that triggers the exocytosis of glucose transporter 4 (GLUT4) to cell membranes — predominantly in striated muscle and adipose tissue — facilitating glucose uptake and lowering blood glucose levels as a result. However, beyond glucose uptake and the broad regulation of blood glucose levels, the IRSP is broadly implicated in promoting the anabolism and modulating the catabolism of every major biomolecular class — polysaccharides, triacylglycerols, proteins, and nucleic acids. It is also responsible for promoting cellular survival, growth and proliferation, while inhibiting apoptosis and autophagy. Its members and metabolic/physiological outcomes are depicted in the Cell Signaling Technology pathway diagram below:
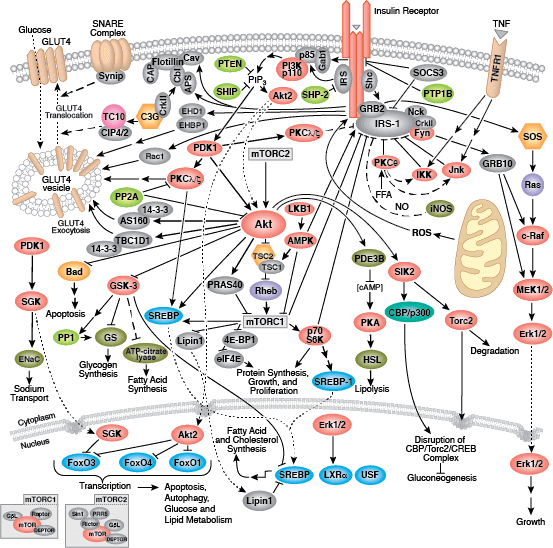
Due to the complexity of the IRSP, there is quite evident potential for numerous molecular pathologies to arise at various hierarchical levels of the pathway; the higher the signaling level at which a molecular lesion develops, the greater the number of metabolic functions and outcomes that may potentially be affected (Boucher et al., 2014). The most common dysfunction stemming from IRSP lesions is non-insulin-dependent diabetes mellitus — more commonly and simply known as type 2 diabetes. While there is somewhat of a stigma surrounding type 2 diabetes — in that it is typically associated with unhealthy lifestyle, poor diet, and insufficient exercise — growing evidence suggests that previous preconceptions may be at least somewhat oversimplified; a greater population of non-obese type 2 diabetics are being recognized, as is the notion that insulin resistance is strictly a cause of obesity versus an effect of obesity (Corkey, 2012; Han and Boyko, 2018; Berumen et al., 2019; Malone and Hansen, 2019; Olaogun et al., 2020). In type 2 diabetic individuals who maintain an otherwise-healthy lifestyle — and/or in individuals where dietary and lifestyle changes are ineffective and/or impossible (e.g. due to mobility issues, etc.) — pharmacological intervention remains important despite societal aversion to drugs; this is only further underscored by projections issued from the International Diabetes Federation that 700 million adults will be living with diabetes worldwide in 2045.
Read more about our interventive arm — reversal of insulin resistance, and our preventive arm — early-detection of insulin resistance biomarkers.
